Abstract
Background
Although the outcome after allogeneic hematopoietic stem cell transplantation (Allo-HSCT) for the patients with acute myeloid leukemia (AML) in complete remission has been improved, the prognosis of patients with active disease is still dismal, generally with 20 - 30% of overall survival (OS) at 2 years. Prognostic value of gene mutations detected by the next generation sequencing (NGS) for this extremely poor group remains to be evaluated.
Methods
A total of 120 patients with AML not in hematological remission who received the first allo-HSCT at our institute between April 2005 and December 2017 were enrolled. For each patient, genomic DNA was extracted from the frozen bone marrow sample harboring leukemic blasts which was preserved at the nearest available date before the initiation of conditioning regimen. Sequencing was performed using TruSight Myeloid Sequencing Panel® on the MiniSeq system (Illumina). Gene variants were detected by in-house analysis pipeline. Charts were retrospectively reviewed on survival, relapse, and non-relapse mortality (NRM). The Kaplan-Meier method was used to assess OS using the log-rank test. Univariate and multivariate analysis were performed to identify potential prognostic factors. The Cox proportional hazards method was used for the multivariate analysis to assess OS. Gray's test and the Fine-Gray test were used to assess the cumulative incidence of relapse (CIR). Competing risks were relapse and NRM.
Results
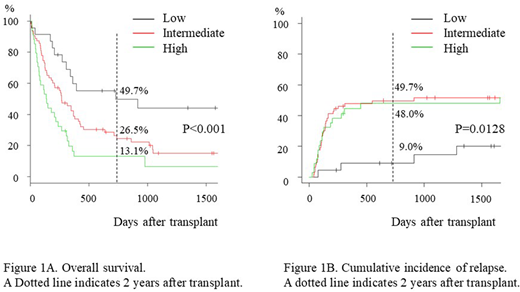
Median follow-up of survivors was 1345 days (235 - 4888 days). Median age at transplant was 51 (range 21 - 71). Grafts were from bone marrow (n = 67, 55.8%), peripheral blood (n=42, 35.0%) and cord blood (n=11, 9.2%). Refined disease risk index (Blood. 2014;123:3664-71) scored high (n=61, 51.3%) and very high (n=58, 48.7%). OS at 2 years of the whole cohort was 27.3% (95% confidence interval [CI], 19.4% - 35.7%). There was no significant difference in OS between patients in primary induction failure and in relapse (OS at 2 years: 26.5% [n=50] vs 28.7% [n=70], p= 0.293). NGS analysis revealed TP53 loss-of-function mutation in 23 (19.2%) patients. Among all detected gene mutations, TP53 mutation was the most powerful predictor of poor OS after allo-HSCT (OS at 2 years: 13.5% vs 30.5% for TP53+ [n=23] vs TP53- [n=97], p= 0.0184). Consistent with previous reports, monosomal karyotype (MK, J Clin Ocnol 2010:26;4791-7) was significantly associated with positive TP53 mutation (13.3% of non-MK vs 37.9% of MK, p=0.006). Of note, all the patients (n=11) positive for both prognostic factors died within 1 year after allo-HSCT, whereas OS of the patients without either factor (n=78) was 33.6% at 2 years. Multivariate analysis on OS revealed MK, TP53 mutation, de novo AML (no prior history of MDS), ECOG performance status score 2 or more, C reactive protein 1.0 mg/dL or more, peripheral blood blast frequency of 1% or more at the initiation of conditioning regimen were independent prognostic factors for poor OS. Among them, to determine the prognostic factors critical for deciding the indication of allo-HSCT, we chose pre-transplant factors which were available around one months before transplant. From this point of view, multivariate analysis revealed independent prognostic factors for poor OS after allo-HSCT including MK (Hazard ratio [HR] 2.05; 95% confidence interval [CI] 1.30 - 3.25, p=0.00217), TP53 mutation (HR 1.72; 95% CI 1.04 - 2.86, p=0.035), and de novo AML (HR 1.67; 95% CI 1.04 - 2.86, p=0.036). Because the HR of these three factors were comparable, a score of 1 was assigned to each factor. OS at 2 years was 49.7%, 26.5% and 13.1% for patients with low (score 0, n=23), intermediate (score 1, n=63) and high (score 2 or 3, n=34) risk, respectively (p<0.001, Figure 1A). CIR was significantly lower in low risk group compared to intermediate and high risk groups (9.0%, 49.7% and 48.0% at 2 years, p=0.0128, Figure 1B), whereas NRM was comparable (41.3%, 32.5% and 41.7% at 2 years, p =0.515).
Conclusions
NGS-analysis revealed the importance of TP53 mutation on transplant outcome of AML in non-remission. The combined score with the information on MK, de novo AML, and TP53 mutation stratified the patients into three groups, including low risk with 50% survival rate, and high risk with shorter survival after allo-HSCT for whom the other strategy might be optimal. Our results would require further validation in larger cohorts.
Harada:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.